Services/
Medical Affairs
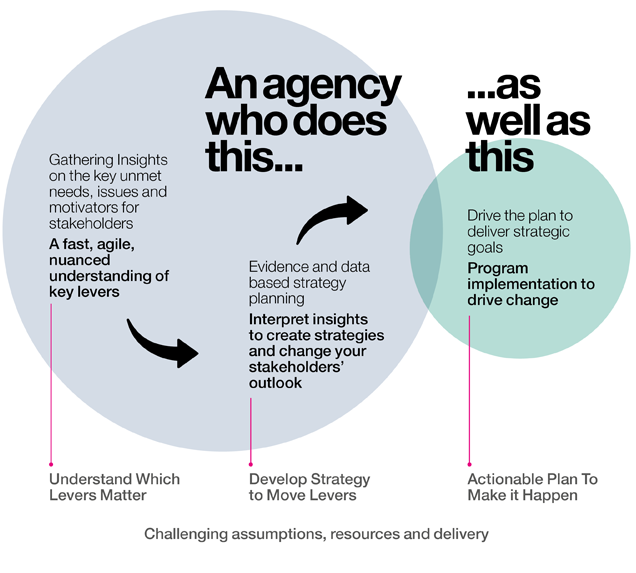
The evolving medical affairs agenda – a ‘New Normal’
Medical Affairs responsibilities have evolved and continue to do so. Pharma faces unprecedented marketplace disruption to the asset commercialisation process. We recognise this and continually innovate to provide best in class solutions to address these challenges. Digital technology and multichannel fluency enhances and accelerates your results across the continuum.
Our goal is to partner with healthcare clients to extract maximum value for each asset in this challenging environment – the new normal. We do this through a deep understanding of your environment, by applying critical thinking to all we do and by proposing solutions that drive customer acceptance and raise the bar for your competitors.
Example key issues:
Increased expectations:
HCP Interaction Not only is there a need to interact with HCPs with greater clinical depth, but clinician expectation is for ever greater digital engagement.
Alignment: the HCP environment
Urgency to understand the key issues and barriers for clinical, regulatory, access, through extended stakeholder relationships
Augmented responsibilities: time constraints
Strategic role requires greater versatility and increased workload to handle changed responsibilities
Regulatory compliance: increased transparency
In all communication and training programmes there is a need to offer technological securement and transparency in compliance. There is a new need for centralised, web-enabled systems to manage and track consistent presentation of messages, and the gathering of insights.
Service Type
Classic medical communication
MedEd Programmes – creation and delivery
Advisory boards – F2F and virtual
Strategic Publications Planning and Publications
Disease Awareness
Scientific Affairs – high science strategy and delivery
KOL Development, Patient Advocacy, Patient Communication/Education, Live Events,
Digital Engagement, eLearning, Gamification, Stakeholder Engagement/Behavioural Change
MSL Optimisation
MSL Optimisation, training and alignment for Market Intelligence – novel tools, platforms, dashboards, KOL digital support items, digital research enhancement
Market Insights
Rapid Market experience – depth enhancement through digital tools. Regional and National.
Digital Engagement
HCP/Association Web Communities, Risk Assessment Tools, Multichannel and Social Media
Clinical and R&D Support
Clinical Trial Communications, Digital engagement, Investigator Web Communities, Investigator meetings, CT Identity/Branding/Comms Internal and External
Strategy
Strategy Development, Portfolio Analysis
Digital across all functions
Training, Comms, Content
Scientific Communications
Internal Comms and Education
Training/ Employee Engagement
National, LOCs, MSLs, all staff
Market Access/Big data/
Reimbursement, Cost Modelling, Risk Assessment Tools, Market Access HEOR






 Navigation Menu
Navigation Menu